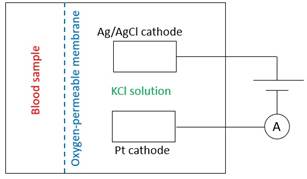
1999B07 Describe how the partial pressure of oxygen in a blood sample is measured using a Clark electrode.
|
Diagramme |
|
|
Components |
· Silver anode · Platinum cathode · KCl salt bridge · 0.6V power source · Ammeter |
|
Reactions |
· Anode: 4 x (Ag + Cl -> AgCl + e-) · Electrolyte: 4 x (KCl + OH- -> KOH + Cl-) · Cathode: O2 + 2H2O + 4e- -> 4OH- |
|
How does it work |
· O2 diffuses down concentration gradient across the plastic membrane into the salt bath · O2 is consumed at the cathode · Hence PaO2 ∝ current · Accuracy ±2mmHg. Delay in analysis may cause reduction in pO2. |
|
Sources of error |
· Non-linear relationship between pO2 and potential difference o But almost linear at 0.6V · Non-linear relationship between potential difference and current o But almost linear at 0.6V · ↑temp -> ↑rate of reactions -> false ↑ o Hence temp control to within 0.1°C · Inaccuracy with time and use o Hence regular two point calibration with pO2 660mmHg and 160mmHg · Blockage of the membrane with blood components -> falsely low result o Hence keep clean · Delay in sampling -> consumption -> ↓pO2 o Hence cool with ice, process immediately |
Feedback welcome at ketaminenightmares@gmail.com