2010A01 Describe
how the chemical structure of local anaesthetic drugs
determines their efficacy and safety.
List:
· Intro
· Lipophilic part
· Link
· Hydrophilic part
· Other: isomerism, ionisation
Intro:
|
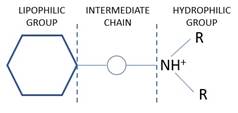
Local anaesthetic structure |
· Lipophilic group · Chain: ester or amide bond and hydrocarbon chain · Hydrophilic group
|
|
Determinant of efficacy |
· Propensity for blockade of neuronal voltage-dependent Na+ channel (VDNaC) |
|
Determinant of toxicity |
· Propensity for blockade of VDNaC in the o CNS (excitation then depression) o CVS (conduction delay, re-entrant arrhythmia, arrest) |
Lipophilic part:
|
Purpose |
· For crossing axonal membrane · For local anaesthetic activity |
|
Structure |
· Usually an aromatic ring |
|
Factors influenced |
· Lipophilicity · Potency · Duration |
|
Examples |
· Ester: o Metabolite PABA may cause hypersensitivity o Excreted unchanged in urine; could accumulate in renal failure · Prilocaine: o Metabolite O-toluidine may cause methaemoglobinaemia o Oxidises Fe2+ in Hb o Significant if >600mg prilocaine given to adult. PABA excreted in urine · Chlorprocaine: o Cl atom ↑rate of ester hydrolysis 3.5x -> ↓risk toxicity |
Link:
|
Ester bond |
· O=COR · Metabolized rapidly in plasma/liver by pseudocholinesterase · ↓peak plasma [LA] -> ↓risk toxicity · Products: aromatic acid + alcohol + tertiary amide · ↑Length alcohol -> ↑potency up to a critical length, then ↑toxicity |
|
Amide bond |
· NHC=OR · Metabolized slowly in liver by amidases (CYP450) · ↑peak plasma [LA] -> ↑risk toxicity · Agent-specific metabolites |
|
Hydrocarbon chain |
· ↑length of chain -> ↑potency |
Hydrophilic part:
|
Purpose |
· For water dissolution |
|
Structure |
· Usually tertiary amine e.g. diethylamine |
|
Determinant of |
· Lipid solubility (LS) ∝ potency ∝ toxicity |
|
Examples: pipecolyxylidides |
Structure-activity relationships: · Chiral due to assymetric carbon (cf. lignocaine) · Length piperidine C-chain ∝ lipid solubility, potency, toxicity, % protein bound Comparisons (reference: procaine) · Methyl (CH3) = mepiv o LS 100x, potency 2x, 77% plasma protein bound (PPB)) · Propyl (C3H7) = ropiv o LS 300x, potency 8x, 94% PPB) o S-ropiv more potent, less toxic, hence always enantiopure · Butyl (C4H9) = bupiv o LS 1000x, potency 8x, 95% PPB o L-bupiv more potent, less toxic, preferred in paeds (expensive) |
Other:
|
Isomerism |
· e.g. S-ropivacaine more potent, less toxic than R-ropivacaine |
|
Ionisation |
· Determines diffusability hence speed of onset o e.g. lignocaine pKa 7.9, 25% unionised at pH 7.4 -> fast onset o e.g. bupivacaine pKa 8.1, 15% unionised at pH 7.4 -> medium onset |
Feedback welcome at ketaminenightmares@gmail.com