2011B12 Outline
the similarities and differences between myoglobin and adult haemoglobin,
explaining the physiological relevance of the differences.
|
|
Haemoglobin |
Myoglobin |
|
Location |
· Erythrocytes |
· Striated
muscle |
|
Structure |
· 4 x haem (protoporphyrin + Fe2+) · 4 x globin (polypeptide chain) · HbA1 97% α2β2 · HbA2 3% α2δ2 |
· Single haem · Single globin |
|
Function |
· O2 carriage o Complex with iron (haem) o ↑CaO2 from ~2mL/100mL to ~20mL/100mL arterial blood · CO2 carriage o CO2 + NH2 <-> NHCOO- + H+ (carbamino) o Also buffers H+ from carbonic acid · Buffer in “ECF” o KHb + H+ <-> HHb + K+ o Imidazole groups of histidine residues |
· Unclear · At rest: ? ↓O2 toxicity · In peak exercise: ?↑gradient |
|
O2 affinity |
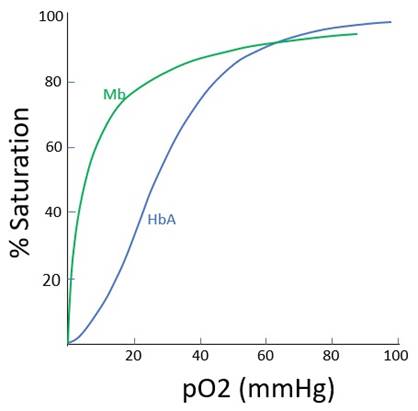
· Moderate, p50 26mmHg |
· Very high, p50 2.8mmHg |
|
Dissociation curve shape |
· Sigmoid (see below) o Multiple subunits with co-operative binding o Also law of mass action |
· Hyperbolic (see below) o Single subunit o No co-operative binding o Law of mass action only |
|
Co-operative binding |
· Binding of one haem to O2 increases affinity of the next haem for O2 · Due to allosteric interactions · Tense (T) closed -> relaxed (R) open state |
· Single subunits, no co-operative binding |
|
Bohr effect |
· R shift due to ↑T, ↑PaCO2, ↑[H+], ↑[2,3-DPG] · Due to allosteric interactions as above. |
· N/A |
|
Haldane effect |
· Capacity to bind or buffer CO2 and H+ is greater in HHb than HbO2 · 70% due to 3.5x ↑ability to form carbaminoHb · 30% due to ↑pKa imidazoles 6.6 to 8.2 |
· N/A |
|
Nephrotoxicity |
· Yes (released upon intravascular haemolysis) |
· Yes (released upon rhabdomyolysis) |
Graph:
Feedback welcome at ketaminenightmares@gmail.com