2007A12 Explain
the physiological principles underlying the use of peritoneal dialysis
in a patient with chronic renal failure with this dialysate solution:
Na+ 132mmol/L, Mg2+ 0.5mmol/L, K+ 0mmol/L,
Lactate 40mmol/L, Cl- 96mmol/L, glucose 2.5%, Ca2+
3.5mmol/L.
List:
· Intro
· Mechanisms
· Special additives
Intro:
|
Definition |
· Exchange between dialysate and blood across a membrane |
|
Purpose |
· Maintain fluid, electrolyte, acid-base homeostasis · Excrete metabolic waste |
Mechanisms:
|
Diffusion |
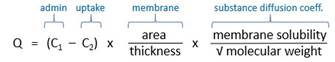
· Movement of solute from high concentration to low concentration o Slow process · Modulated by Gibbs-Donnan effect (?) o Proteins are anionic, membrane-permeable and attract small cations e.g. Na+ o (Not sure how this is relevant) |
|
Osmosis |
· Movement of solvent from low osmolality to high osmolality o Dialysate 400mOsm and hypertonic (mainly due to glucose) o Plasma 290mOsm, by definition isotonic o Osmolarity: number of osmotically active particles per L § ≈= 2 x Na+ + urea + glucose o Tonicity: effect of a solution on cell size (hypertonic -> swelling) |
|
Solvent drag |
· Movement of solute due to bulk flow of solvent · May allow movement of a substance against a concentration gradient |
Special additives:
|
Lactate |
· Metabolized by liver to produce HCO3- · i.e. buffer for H+ |
|
Glucose |
· Large amount · Can cause hyperglycaemia, obesity, insulin resistance |