2019A04 Describe the ways in which carbon dioxide is carried in the blood.
List:
· Key numbers
· Carriage
· Variable CO2 affinity
Key numbers:
|
|
Arterial |
A-V difference |
|
As HCO3- |
90% |
60% |
|
As carbamino compound |
5% |
30% |
|
As dissolved CO2 |
5% |
10% |
|
|
|
|
|
Tension |
40mmHg |
6mmHg |
|
Content |
48mL/100mL |
4mL/100mL |
Carriage:
|
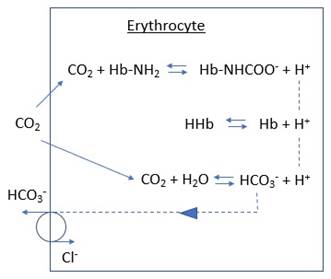
As HCO3- |
· Location: o Haemoglobin (140g/L, 39 imidazoles per molecule) o Plasma proteins (70g/L, 13 imidazoles per molecule) · Purpose: o Maintain partial pressure gradient for CO2 from tissues into capillary blood · Mechanism: o CO2 + H2O < -> H+ + HCO3- (catalysed by carbonic anhydrase in RBC) o KHb + H+ <-> HHb + K+ (buffering by imidazole groups of histidine residues) o HCO3- exchanged for Cl- at membrane (Hamburger effect, facilitates above reactions) |
|
As carbamino compound |
· Location: same · Purpose: same · Mechanism: NH2 + CO2 <-> NHCOO- + H+ |
|
As dissolved |
· Amount dissolved = PaCO2 x k (Henry’s law) · Solubility coefficient k: 0.03mmol/L/mmHg at 37°C · Note ↑temp -> ↓solubility -> ↑PaCO2:CaCO2 |
|
Illustration |
|
Variable CO2 affinity:
|
Haldane effect |
· Higher uptake of CO2 and H+ in HHb c.f. HbO2: o 70% of increment due to 3.5x ↑carbamino formation o 30% of increment due to ↑buffering (imidazole pKa 6.6 -> 8.2) |
|
Isohydric buffering |
· For each mol O2 unloaded by Hb, 0.7mol H+ can be added without ↓ plasma pH · Due to Haldane effect |
Feedback welcome at ketaminenightmares@gmail.com